On The Determination Of Cracking Limits In Cryopreserved Cat Brains
by Dr. Yuri Pichugin
(The following was compiled from several separate reports of various phases of the work, the last received in May of 1998. Any flaws in editing or translation are our own. Reminder: There were no experiments done on live animals. The cats that were anesthetized and then euthanized would have been euthanized in any case.)
I work with cat heads in order:
- to determine cracking limits;
- to study a mechanism of cryodamage by cracking;
- to compare an anti-cracking effectiveness of CPAs (glycerol, DMF, and another);
- to find the most effective procedure of CPA perfusion and reperfusion;
- to verify the most important results of the cat head experiments on sheep head and skulls; Also, to verify a correction of Rubinsky’s equation and, maybe, to perfect it;
- to find the optimum CPA and method of cryopreservation and to verify it using cat brain slices;
- to perform more perfect calculations of cracking limits for human brains using the results of the determination of the cracking limits of the cat and sheep brains.
Transition from the work with sheep brains to the work with cat brains was conditioned by greater accessibility cats than sheep and a possibility to work with the cat brains in short order after death of the animals that it is especially necessary to work brain slice culture. The purposes of this work here reported was to study the effect of freeze-thawing modes on the morphological structure of the native cat brains and the cat brains perfused with cryoprotectant agents (CPAs) DMF (dimethyl formamide) and glycerol.
MATERIALS AND METHODS
General methods for all the cat brains are described here only. Cats (both cats and tom-cats) weighing 2.6-3.6 kg were used in the work. Firstly the animals were anesthetized by ether. Then 200 mg/kg hexanalum was injected intramuscularly. In 20-30 mins after that, intravenous injection (150 mg/kg) was performed for deep general anesthesia. Anesthetized cats were killed by rapid cutting of the throat with an extremely sharp knife, if their heads were not used for CPA perfusion. For CPA perfusion, anesthetized cats were killed by cutting of the jugular veins only. In 5 mins before that, heparin (700 units/kg) was injected into the jugular veins.
The washout cat heads from blood was performed by injection of 0.5 L of Tyrode’s solution with heparin (10 units/ml) via the carotid arteries cannulated at the level of the second verterbra. A period of time from a moment of death of a sheep to the beginning of washout of head tissues from blood was practically absent. The rate of feeding washed solution was about 40 ml/min. The cat heads were then perfiised by low concentrations of DMF {20% DIVIF on Tyrode’s solution) or 20% glycerol on T. s. All solutions were prepared using analytical grade chemicals. Temperature of the solutions was 25 C (room temperature). After perfusion, the cat heads were cut at the level of the second vertebra.
To obtain faster rates of freeze-thawing, all tissues and bones around the cat skulls were moved away. Then the heads or skulls were wrapped up in an aluminum foil and rapidly plunged directly into LN2. Temperature was measured by copper – constantan thermocouples. Thawing of the skulls was performed using a water bath at + 40 C. Washout of the whole brains from CPAs (reperfusion) after thawing was not performed. After thawing, the brain was removed for examination. Pieces of the frontal lobes of the brain were used for histological preparations. Pieces of the back part of cerebral hemispheres were used for determination of CPA concentrations. Determination of glycerol concentration in head tissues (the white and grey matter of brain, the mastication muscle, and the tongue) was carried out by the method previously described in The Immortalist 1994, v. 25, No. 8, p.5. (I’ll write a special article on determination of CPA concentrations in tissues and cells.) The washout of cat brain pieces were not carried out. The tissues with CPA were fixed in 10% neutral formalin with addition of CPA.
RESULTS AND DISCUSSION
(Before description of freeze-warming modes, I give an information about average rates using Figs. 1,2 as an example. Figs. 1,2 concern my article in The Immortalist 1997, No. 3/4, p. 48-50 (about the sheep skulls). I am sorry, I have written in Figs. 1,2 “sheep heads” instead of “sheep skulls”. Comparing the average rates in the Figs. with the ones in that article (p. 49), one can see that the average rates in Figs. 1,2 more correctly reflect the curve of freeze-warming. I’ll therefore give an information about freeze-warming modes using merage rates on approximately linear regions of the curves of freeze-warming. All data on the freeze-warming modes were processed by PC (Pentium 133) and were presented in the form of the graphs. That is very helpful for my work.)
In the beginning of the work, the maximum cooling and warming rates of cat heads and skulls were found. Also, the present of macrocracks in those thawed cat brains was verified.
Here is a summary of the report:
1. 1 A native head (295 g) of a cat (3.3 kg) was frozen over the surface of LN2 and warmed at +40 C using an aqueous bath. The first thermocouple (tcl) which was located in the palate showed the cooling rates of 0.51 C/min from -5 C to -106 C, 0.43 C/min from -106 C to -145 C, and about 0.27 C/min to -196 C. The second thermocouple (tc2) which was located at a distance about 5 cm from the foremen of the second vertebra ( about in the middle of the brain) showed the cooling rates of 0.67 C/min from -5 C to -10 C, 0.51 C/min from -100 C to -145 C, and about 0.30 C/min to -196 C. The warming rates were 8.8 C/min from -196 C to -50 C for tcl and 8.4 C/min for tc2 in the same temperature region.
1.2 The same cat head was then frozen by a direct immersion into LN2 with the rates 27.0 C/min from -13 C to -116 C, 9.14 C/min from -116 C to -166 C, 2.05 C/min to -196 C for tcl and 20.3 C/min from -6 C to -151 C, 5.50 C/min from -151 C to -186 C for tc2. The head was warmed at +40 C with the rates 21.4 C/min from -196 C to -50 C, 0.94 C/min from -50 C to -18 C, 0.30 C/min to 0 C for tcl and 16.5 C/min from -196 C to -37 C, 1.0 C/min from -37 C to -7.5 C, 0. 14 C/min to 0 C for tc2. The brain of this cat head was very strongly cracked. It was not fixed in formalin and photographed.
2. Pilot experiments on determination of freeze-warming modes of a cat head perfused with 20% glycerol and its skull. The cat weighed about 4.0 kg, its head weighed 307 g, and its skull was 53 g. The thermocouple 1 and 2 had the same location as in the head of experiment 1. 1.
2.1 The head was frozen by immersion into LN2 with the rates 3.5 C/min from -12 C to -176 C for tcl and 4.6 C/min in the same temperature zone for tc2. The cat head was thawed at +40 C with the rates 14.1 C/min from -196 C to -80 C, 4.0 C/min to -24 C for tcl and 8.7 C/min from -196 C to -65 C, 3.0 C/min to -24 C for tc2. {These modes were presented in Fig. 3.4 which had been sent you. I am sorry, I have written in the Fig. 5 “a native cat head” instead of “a cat head perfused with 20% glycerol”}. The freeze-warming rates for the native cat head of experiment 1.2 were slower than the ones for the glycerolized head of this experiment.
2.2 The skull of the cat head was frozen only to -90 C by immersion into LN2 and thawed at +40 C. The thermocouple 1 was located on the bottom part of the skull under 1-2 mm of the tissue. Tc2 was located at a distance about 2.5 cm from the foremen of the atlas. The location of the probes was used for all the cat skulls later on.
2.2. 1 The cooling rates were 22.7 C/min from -4 C to -72 C for tcl and 24.7 C/min from -12 C to -65 C, 5.5 C/min from -65 C to -88 C ibr tc2. When temperature of tcl reached -72 C, the skull was rapidly lifted from LN2, then rapidly plunged into LN2, and again lifted from it so as not to reduce temperature in the brain lower -90 C. When temperature of tc2 reached -90 C, the skull was placed at the level over LN2, where temperature was – 90 Q and kept for 15-20 mins. Freezing the skull was performed into a high cylindrical vacuum flask ( high of the glass vessel is 1200 cm, its diameter is 100 cm) with the exactly determined temperature levels over the surface of LN2. The skull was warmed at +40 C. The warming rates were 37.6 C/min from -90 C to -36 C, 7.4 C/min from -36 C to -12 C for tcl and 15.3 C/min from -90 C to -37 C, 6.3 C/min from -37 C to -18 C for tc2.
2.2.2 A duplication of the experiment 2.2.1 with the same cat skull. The freezing rates were 73.8 C/min from -5 C to -36 C, 21.1 C/min from -36 to -97 C for tcl and about 2.0 C/min from -5 C to -12 Q 33.3 C/min from -12 C to -37 Q 62.2 C/min from -37 C to -65 Q 27.0 C from -65 C to -80 Q 6.3 C from -80 C to -90 C 1br tc2. The warming rates were 23.8 C/min from -90 C to -24 Q 4.8 C/min from -24 C to -4 C for tcl and 20.7 C/min from -90 C to -30 Q 5.7 C/min from -30 C to -18 Q 1.24 C/min from -18 C to 0 C for tc2. The duplication showed a rather bad reproduction of the freeze-warming modes using a manual control but not an automatic programmed control. {To reproduce by hands freezing modes of rat hearts is still more difficult because a rat heart weighs about 1 g.)
2.3 The same skull was also two times frozen to -196 C and warmed.
2.3.1 The cooling rates were 108.0 C/min from -5 C to -50 C, 22.5 C/min from -50 C to -176 C, 5.0 C/min from -176 C to -196 C for tcl and 11.1 C/min from -5 C to -18 C, 53.9 C/min from -18 C to -123 C, 24.4 C/min from -123 C to -176 C, 6.10 C/min from -176 C to -196 C for tc2. The warming rates were 114.0 C/min from -196 C to -80 C, 30.0 C/min from -80 C to -42 C, 8.72 C/min from -42 C to -6 C for tcl and 49.1 C/min from -196 C to -65 C , 20.4 C/min from -65 C to -43 C, 5.28 C/min from -43 C to -10 C for tc2.
2.3.2 A duplication of experiment 2.3. 1. The cooling rates were 26.2 C/min from -4 C to -176 Q 5.76 C/min from -176 C to -50 C, 9.5 C/min from -50 C to -12 C 1br tcl and 44.5 C/min from -196 C to -50 C, 7.7 C/min from -50 C to -18 C for tc2. The method of the rapid immersion of the skull into LN2 had a rather good reproduction. The brain of this cat head was very strongly cracked as well as the first cat brain ( the experiment 1). It was not fixed in formalin and photographed.
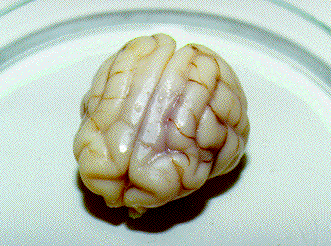
3. Freezing a glycerolized cat head to -90 C. The glycerolized head (274 g) of the cat (3.8 kg) was frozen over the surface of LN2 to -90 C and warmed at +40 C. The cooling rates were 3.0 C/min from -16 C to -80 C [(16 – 80) – used as an abridgement below] and 0.9 C/min (80 – 90) for both tcl and tc2. The warming rates were 3.8 C/min (90 – 32), 1.3 C/min (32 – 18), 0.38 C/min (18 – 0) for tcl and 3.3 C/min (90 – 37), 1.2 C/m in (37 – 18), 0.25 C/min (18 – 0) for tc2. The photos 1. 1 – 1. 3 show the brain of the cat head after warming and fixation.
Photos below – 1 shows the upper surface of a cat brain, a photo .2 shows the lower surface of the cat brain, and a photo .3 demonstrates the brain cutting into 2-3 longitudinal sections for determination macrocracks inside brains. All brains are on a Petri cup. The back parts of cerebral hemispheres from CPA-perfused brains were cut after thawing for determination of CPA concentrations. Then the brains were fixed in 10% formalin. After fixation, the brains were photographed (the top and the bottom), and pieces of the frontal lobes were cut for histological preparations. Only after that, brains were cut into the sections which were also photographed. The top of the photos is that where the numbers of photos had been written.
The photo 1. 1 shows the top of the glycerolized brain on its front side. There are no macrocracks. The large central fissure between cerebral hemispheres was very well observed. The photo 1.2 shows the bottom part of the brain on the front side too. There are no macrocracks too. The back parts of the hemispheres were taken for determination of CPA concentration.
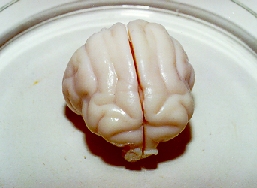
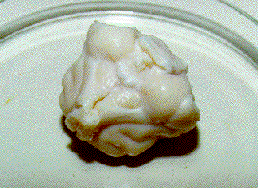
The photo 1.3 demonstrates the three longitudinal sections of the brain without any macrocracks and macroruptures. The part of the front lobes was taken ibr the histological examination of microcracks. The “cavities”, which are observed on the left piece, are not cracks. The macrocracks have the characteristic appearance (see below). That is the convolution and the other bounds of the macrostructures of the brain. On the right piece, the white spots (like white colouring) may be observed. However that is not any colouring but is patches of light which were resulted from a photo-flash. The photos 1. 1-1. 3 showed one of the best cryopreserved brains.
4. Freezing a native head (314 g) of a cat (3.8 kg) to -90 C.
The head was washed out from blood but was not perfused with any cryoprotective agent. The freezing was carried out by immersion of the head into methylcellosolve bath cooled to -90 C. Thawing of the head was performed using the water bath at +40 C. Tc2 was used in this experiment only. The cooling rates were 1.3 C/min (8 – 64) and 0.33 C/min (64 – 90). The warming rates were 3.8 C/min (90 – 19), 1.1 C/min (19 – 10), and 0.2 C/min (10 – 0).
The photos 2.1 – 2.3 display the fixed brain of the cat head. This brain (the photo 2. 1) has the dura matter which was usually ablated. The photo 2.2 shows the lower part of the brain on the side of the back part. In all the cases, the cat brain was taken out from its skulls without its cerebellum because the cat cerebellum was located into the special compartent of its skull in contrast to the sheep cerebellum. Therefore to take out the cat brain together with its cerebellum from its skull is very complicated and difficult. A foremen from the tip of the thermocouple 2 is seen in the photo 2.2. The photo 2.3 shows that macrocracks are absent inside the brain as well as on the upper and lower surfaces. On the upper left section, the central fissure was observed but it is not a crack. The brain even without glycerol was well cryopreserved as well as the glycerolized brain in the experiment 3.
5. To test the presence or absence of macrocracks in harder conditions, a cat brain was frozen using faster rates but to -90 C too. The cat weighed 2.2 kg. Its skull weighed 49 g. The native skull was frozen using direct immersion into LN2 but to -90 C as the same way as the skull in experiments 2.2. The cooling rates were 32.4 C/min (0 – 80), for tcl and 22.6 C/min (10 – 50), 8.6 C/min (50 – 78), 1.2 C/min (78 – 90) for tc2. The warming rates were 27. 0 C/min (90 – 65), 19.1 C/min (65 – 24), 6.3 C/min (24 – 0) for tcl and 13.0 C/min (90 – 18), 2.8 C/min (18 -4) for tc2. The rates of this experiment were approximately comparable with the rates of experiments 2.2. However the rates were much faster than the rates of experiment 3 and 4.
The photos 3.1-3.3 demonstrate this fixed brain without evident macrocracks. However the lower slice of the brain in the photo 3.3 possibIly has a rupture as prolongation of the central fissure. The final conclusion about cracking may be made only after examination of the histological preparations of this brain. The brain was not washed out from blood but its blood vessels were not observed because haemoglobin lost its colour during 1-1.5 month of keeping in formalin.
6. Freezing a glycerolized head (328 g) of a cat (3.2 kg) to -196 C. The cooling rates were 4.0 C/min (24 – 183), 0.73 C/min (183 – 196) for tcl and 4.7 C/min (16 – 183), 0.65 C/min (183 – 196) for tc2. The warming rates were 9.3 C/min (196 -42), 3.5 C/min (42 – 30), 0.84 C/min (30 – 0) for tcl and 8.0 C/min (196 – 50), 4.0 C/min (50 – 37), 0.71 C/min (37 – 0) for tc2. The rates were wry similar to the rates of experiment 2. 1.
The photos 4.1-4.3 display the fixed brain of this experiment. The photos 3.1-3.2 show that the brain was wry well washed out from blood and has no macrocracks on its surface. However the lack of the cracks on the surface of the brain does not signify the absence of the macrocracks inside the brain. So the photo 3.3 demonstrates a few macrocracks on the brain sections. The cracks are shown by the arrows. The top part of the brain was broken down into the two pieces (hemispheres) along the central fissure (on the lower part of the photo 3.3). On the upper left part of the photo, the middle part of the brain has a large crack ( or a rupture) as prolongation of the central fissure. The section was almost broken down into two pieces. On the upper right part of the photo, the lower part of the brain also contains the same rupture. Thus, the given mode of freeze-warming does not cryopreserved the glycerolized cat brain from macrocracks.
7. Freezing a native skull (77 g) of a cat (3.7 kg) to -196 C. The cooling rates were 33.1 C/min (0 – 88), 14.6 C/min (88 – 144), 9.6 C/min (144 – 176), 2.1 C/min (176 – 196) for tcl and 32.0 C/min (12 123), 13.8 C/min (123 – 176), 2.7 C/min (176 – 196) for tc2. The warming rates were 133 C/min (196 – 115), 50.9 C/min (115 – 58), 21.5 C/min (58 24), 4.9 C/min (24 – 0) for tcl and 33.1 C/min (196 – 56), 12.4 C/min (56 – 24), 4.7 C/min (24 – 12) for tc2. The rates were in general slightly slower than the ones of the experiment 2.3 because the weight of this skull (77 g) was less than that skull (53 g).
The photos 5.1 – 5.3 show the brain after thawing and fixation. The photo 5.1 displays that hemoglobin of the unwashed brain have lost its colour 30-50 per cent. There are no macrocracks on the upper surface of the brain. On the lower surface, the small crack is present in the right part of the brain. The photo 3.3 demonstrates the obvious macrocracks, but the brain does not break down into pieces. Also, the cracks are located across the central fissure but not along it.
8. Freeze – warming a native skull (47.4 g) of a cat (2.7 kg) with slower rates at the temperature zone from -90 C to -196 C.
The freeze-warming of the skull was performed into the high vacuum flask (see experiments 2.2). In the beginning of freezing, the skull was rapidly immersed into LN2 1br about a minute. Then the skull was lifted from LN2 and again plunged into LN2 as long as tc2 showed -80 C. After that, the skull was kept at – 90 C over LN2 for few minutes and then frozen with a slow rate to -196 C. The cooling rates were 84 C/min (15 – 88), 8.0 C/min (88 – 65, warming), 17 C/min (65 – 100), 8.0 C/min (100 – 90, warming), 0.2 C/min (90 – 107), 0.3 C/min (107 – 166), 0.87 C/min (166 – 196) for tc1 and 18.2 C/min (5 – 50), 5.4 C/min (50 – 56), 17.1 C/min (56 -78), 5.0 C/min (78 – 88), 0.19 C/min (88 – 107), 0.28 C/min (107 – 166), 0.87 C/min (166 – 196) for tc2. The warming rates were 0.96 C/min (196 – 175), 0.32 C/min (175 – 80) for tcl and tc2. After warming the skull to -80 C over LN2, it was warming using a water bath at +40 C with the rates 48 C/min (80 – 50), 13.1 C/min (50 – 18) for tcl and 35 C/min (80 – 65), 11.0 C/min (65 – 18) for tc2.
The photos 6.1 – 6.3 show this fixed brain with visible blood vessels but dissociating hemoglobin. The upper surface of the brain has no macrocracks (Ph. 6. 1). The photo 6.2 shows the back part of the brain with a foremen from tc2 but without macrocracks. However few cracks were found inside the brain (Ph. 6.3). This brain looked slightly better than the brain of the experiment 5, although even the such slow rates at the zone (196 – 90) cannot preserve the native brain against the macrocracks.
9. Freeze-warming a native head (212 g) of a cat (2.5 kg) with slower rates at the zone from o C to -90 C.
The cooling rates were 6.2 C/min (18 – 144), 2.27 C/min (144 – 181), 0.39 C/min (181 – 196) 1br tcl and 6.9 C/min (18 -148), 1.83 C/min (148 – 181), 0.35 C/min (181 – 196) ibr tc2. The warming rates were 16.0 C/min (196 – 106), 7.4 C/min (106 – 50), 2.8 C/min (50 – 12) for tcl and 14.1 C/min (196 – 100), 7.4 C/min (100 – 50), 2.78 C/m in (50 – 12) for tc2.
The photos 7.1-7.3 show this fixed brain with the clearly visible blood vessels. Hemoglobin was well remained during two weeks from the beginning of brain fixation to a moment of the photographing. The morphological structure of this brain is very like the one of the brain in the experiment 8. Thus, the freeze-warming modes no visible influence on the native brain in comparison with that experiment.
10. Freeze-warming a cat head (400 g) perfused with DMF.
The thermocouple 2 was used for this experiment only. The cooling rates were 4.6 C/min (0 – 18), 18.2 C/min (18 – 128), 6.58 C/min (128 – 177), and 1.12 C/min (177 -196). The warming rates were 15.2 C/min (196 – 73), 6.7 C/min (73 – 43), and 2.6 C/min (43 – 10). The rates of this experiment were faster than the ones of the like experiment 6 with glycerol.
The photos 8.1-8.3 show the fixed brain. There were no macrocracks on the upper surface of the brain (Ph. 8. 1). The photo 8.2 displays mainly the back part of the brain with the macrocracks, but the lower surface has no visible cracks. The middle part of the brain had more numbers of the macrocracks than the one of the glycerolized brain of the experiment 6. Thus, this brain looked worse than that brain in general.
11. Freeze-warming a cat skull (61,4 g) perfused with DMF.
The cooling rates were 10.8 C/min (24 – 50), 21.3 C/min (50 -178), 3.13 C/min (178 – 196) for tcl and 17 C/min (6 – 176), 3.85 C/min (176 – 196) for tc2. The warming rates were 57 C/min (196 – 133), 22.7 C/min (133 – 80), 13.1 C/min (80 – 42), 4.6 C/min (42 – 12) for tcl and 40 C/min (196 – 162), 23.5 C/min (162 – 78), 13.5 C/min (78 – 50), 4.7 C/min (50 – 18) for tc2.
The photos 9.1-9.3 show the fixed brain. This CPA-perfused brain looked (in some reason, ?) worse than even the native brain in the like experiment 7. So even the upper surface of the brain had a large crack across the central fissure (Ph. 9. 1). The lower surface had few macrocracks too (Ph. 9.2). A slice of the brain broke down into pieces (P h. 9.3).
12. Freeze-warming a cat skull (61,8 g) perfused with DMF and warming it with slower rates.
The cooling rates were 44 C/min (5 – 80), 19.9 C/min (80 – 176), 4.5 C/min (176 – 196) for tcl and 35 C/m in (24 – 176), 2.3 C/min (176 – 196) for tc2. The rates were faster than the rates in the experiment 11. The warming rates were 0.25 C/min (196 – 155), 0.5 C/min (155 – 133), 1.0 C/min (133 – 72) 1br both tcl and tc2. Then the skull was warming on air at +24 C with the rates 5.34 C/min (72 – 42), 2,56 C/min (42- 10) for tcl and 4.15 C/min (72 – 73), 2.03 C/min (37 – 10) for tc2.
This brain looked rather better than the brain of preceding experiment (Phs. 10. 1-10.3). The upper surface of the brain had no macrocracks (Ph. 10.1). The lower surface had a crack only (Ph. 10.2). The middle part of the brain contained macrocracks (Ph. 10.3) like the brain of the experiment 8 (Ph. 6.3). The slower warming made somewhat better cryopresenation of the brain.
Thus, some over-usual, remarkable properties of DMF have not been found in the present for the hard conditions of brain cryopresentation (low CPA concentrations and fast rates). The next series of experiments will be devoted to comparison of cryoprotective activity of DMF with one of glycerol for easier conditions of brain cryopreservation (slower rates and higher CPA concentrations).
HISTOLOGICAL EVALUATION OF CAT BRAIN EXPERIMENTS 3-6 AND 12
As reported previously, macrocracks were absent in the cat brains of the experiments 3-5 (freezing to -90 C) and so histological preparations were made in order to examine the presence of microcracks. Histological preparations of the experiments 6 and 12 (freezing to -196 C) were made to determine shape of microcracks and extent of cell damage of the cat brain tissues that were frozen with glycerol or DMF.
The microcracks were present in all the histological preparations of all the experiments 3-5 in spite of the absence of the macrocracks. However the shape of these microcracks differed from the shape of the microcracks of the untreated sheep brain tissue frozen to -196 C (The Immortalist 1996, v. 27, No. 7/8, p. 31) and the CPA-perfused sheep brain tissues frozen to -196 C (The Im. 1997, v. 28, No. 3/4, p. 48-50). Apparently, the microcracks which were formed in the cat brain tissue at freezing to -90 C did not result from thermo-mechanical tensions because this type of damage is characteristic for temperature below -90 C. They may be a result of a kind of ice crystallization. It is therefore better to use words “cavities” or “ice holes” for the name of “the microcracks from freezing to -90 C”.
The third experiment. Freezing the glycerolized cat head to -90 C. The histological preparations of the brain tissue were stained by the Nissl method (Nl) and with hematoxylin and easin (h.-e.). All micrographs had a magnification 350x (h.-e.) and 700x (Nl).
The typical appearance of the cavities (or the ice holes) is shown in the micrographs 1,2 (Figs. 1,2 the white matter) and in Figs. 2,3 (the grey matter). The cavities in the white matter had a more round shape than the ones in the grey matter. The ice holes occupied approximately 20% of the grey matter and 40% of the white matter. Fig. 5 shows the typical appearance of the grey matter without the cavities. Figs. 6,7 demonstrate the stellate neurons. The neurons had irregular shapes and stained nuclei. The nucleoli were faintly seen. The pyramidal neurons have a like appearance (Figs. 8,9). Disintegrating nerve cells were very rarely met (Fig. 9).
The fourth experiment. Freezing the native cat head to -90 C.
The cavities in the white matter of the brain tissue had some greater sizes than the cavities in the white matter of the brain tissue of the experiment 3 (Fig. 10). The grey matter also contained the cavities (Figs. 11, 12). The ice holes occupied approximately 30% of the grey matter and 50% of the white matter. The neurons looked worse than the ones in the experiment 3. They more strongly uptake the stain. The nucleoli were not seen (Figs. 13, 14 the stellate neurons, Figs. 15, 16 the pyramidal neurons.)
The fifth experiment. Freezing the native cat skull to -90 C.
Distribution and sizes of the cavities in the brain tissue of this experiment were very like ones of the experiment 4. Figs. 17, 18 shows the typical cavities in the white matter. Figs. 19, 20 demonstrates the typical cavities in the grey matter. The neurons looked slightly worse than the ones in the experiment 4. A great number of the destroying neurons were found (Figs. 21, 22 the stellate neurons, Fig. 23 the pyramidal neurons).
The sixth experiment. Freezing the glycerolized cat head to -196 C.
The main feature of the histological picture of the cat brain tissue of the experiments 6 and 7 was a combination of the cavities with the microcracks from thermo-mechanical tensions. The white matter contained the cavities only (Figs. 24, 25) which looked as well as the cavities in the white matter of the experiment 3. The ice holes occupied approximately 40% of the white matter too. Fig. 26 shows the region of the white matter without the cavities. The grey matter had the typical microcracks side by side with the cavities (Figs. 27, 28). Usually, the microcracks were finished at the border of the grey matter with the white one (Fig. 29). The ice holes occupied approximately 20% of the grey matter. About 10% of the grey matter was occupied by the microcracks. Therefore this cat brain had only few microcracks inside. Fig. 30 demonstrates the region of the grey matter without the cracks and cavities. Some accumulations of fluid were observed near the neurons. The neurons looked very like the ones of the brain tissue of the experiment 3 (Figs. 31, 32 the stellate neurons, Figs. 33, 34 the pyramidal neurons).
The twelfth experiment. Freeze-warming the cat brain perfused with DMF.
This experiment was chosen for histological evaluation as the best of the experiments 10-12 with the use of DMF as cryoprotectant agent (CPA). Morphology of the brain tissue was very like morphology of the brain tissue of the experiment 6. There were the like appearance and a number of the cavities in the white matter (Fig. 35). The grey matter also had the typical microcracks (Figs. 36, 37). However they were some more often met and had some greater sizes (Figs. 38, 39). Therefore this cat brain contained a greater number of the macrocracks than the brain of the experiment 6. But in contrast to that experiment, the neurons practically have no accumulation of fluid near their bodies (Fig. 40 and 36-39). The structure of the neurons was also like the one of the experiment 6.
Thus, the cat brain tissues frozen to -90 C contained only the cavities which did not convert into any macrocracks or macroruptures. The cat brain tissues frozen to -196 C contained not only the cavities but also the microcracks which converted into few macrocracks. Glycerol and DMF even in low concentrations favoured some better cryopreservation of the neurons however they looked pretty bad.
A comparison of the results of the cat experiments 3-5 with the freezing of the native sheep head to -90 C (The Immortalist, 1996, v. 27, No. 7/8, p. 31-32) shows that, likely, sheep’s brain tissues are more cryoresistant than cat’s brain tissues because they did not contain the cavities after freezing. However the sheep brain was frozen with the slower rate 20 C/hour (0.33 C/min) than the cat brains. Only in the cat experiment 4, the cooling rate was nearer that rate (1.3 C/min from -6 to -64 C and 0.33 from -64 to -90 C).
Comparing the cat experiments 6 and 12 with freezing the sheep brains using the faster rate (about 10 C/min, The Immortalist 1997, v. 28, No. 3/4, p. 48-50), one can note that the sheep brain tissues did not contained any cavities or ice holes but had the stronger damages by a great number of the micro- and macrocracks. Maybe, there were different rates of growth of the microcracks for sheep and cat brain tissues. The sheep brain tissues could have a faster rate of growth of the microcracks because they cut the neurons. The cat brain tissues could have a slower rate of growth of the microcracks and cavities because the cut neurons were very rarely met. The neurons of the cat brains could be moved aside by the slowly growing microcracks or cavities. Apparently, a deformation of the frozen cat brain tissues was more plastic than for the frozen sheep ones.
FREEZE-WARMING CAT BRAINS AT THE RATE 1 C/MIN AT THE ZONE FROM -90 C TO -196 C.
Using Rubinsky’s equation (Cryobiology, v. 17, pp. 66-73 (1980)), the rate of freeze-warming at which cracking of the cat brains should not occur have been calculated like the calculation for a human brain (The Immortalist 1997, v. 28, No. 3/4, p. 50). {In the article in The Immortalist, all the radiuses (R) have to be in square. I had sent so, but, maybe, during a reformation of my e-mail message for the article “square” as 2 over R has been lost.}
An average weight of the cat brain was 21 g. The radius of the brain sphere was 1.71 cm. The rate, faster which strong cracking of the cat brain may be, is 27 C/min. The rate, slower which no cracking of the cat brain may be, is 2.7 C/min. We chose the rate 1 C/min at which cracking of the cat brain should not occur according to Rubinsky’s equation. However, cracking occurred.
A numeration of experiment and photos was continued from the part 1 of the report.
13. Freezing a glycerolized cat skull (46.6 g).
For this experiment and the experiment 14, freeze-warming was carried out into the high cylindrical vacuum flask (see the experiment 2.2.1).
The cooling rates were 37.5 C/min from 0 C to -30 C (0 – 30), 27.3 C/min (30 – 80); 1.5-1.0 C/min (90 -196) for all the subsequent experiments for the thermocouple 1 (tc1) and 12.8 C/min (5 – 80); 1.0 C/min (90 – 196) for all the subsequent experiments for tc2. The warming rates were 1.5-1.0 C/min (196 – 80) for all the subsequent experiments, 11.3 (80 – 0) C/min for tc1 and 1.0 C/min (196 – 80) for all the subsequent experiments, 5.3 C/min (80 – 5) for tc2. The cat brain weighed 18 g.
The photos 11.1 – 11.3 show the brain of the cat skull after warming and fixation. There are no macrocracks on the upper surface of the brain (Ph. 11.1). The lower surface had a large crack on the left lower part of the brain and a small crack on the left upper part of the brain (Ph. 11.2). However the cracks were superficial and did not carry inside the brain as it is seen in the photo 11.3 (the left upper piece is the lower part). There were no macrocracks inside this brain (Ph. 11.3).
14. A duplication of experiment 13.
The glycerol perfusion of the cat head of this experiment was performed unsuccessfully. However this was cleared up after thawing the skull and the determination of glycerol concentrations in the brain tissue. The content of glycerol was two time lower than in the experiment 13. The cat skull weighed 53.5 g and its brain weighed 20.2 g. The cooling rates were 34.3 C/min (0 – 30), 25.2 C/min (30 – 80) for tc1 and 10.3 C/min (5 – 80) for tc2. The warming rates were 27.8 C/min (80 – 0) for tc1 and 10.0 C/min (80 – 5) for tc2.
The photos 12.1 – 12.3 show the fixed brain. The brain was well washed out from blood. The upper surface of the brain has no macrocracks and look well (Ph. 12.1). The lower surface had a small crack on the middle left part of the brain (Ph. 12.2). 6-8 macrocracks were found inside the brain (Ph. 12.3). Most likely, the greater number of the cracks resulted from the lower glycerol concentration. But also another cause may be. So, in time of moving dawn or up a container with a cat skull into the high cylindrical vacuum flask, joggles of the skull against the walls of the flask can initiate the macrocracks during freeze-warming at the zone from -90 C to -196 C. To verify the presumption, a new chamber for controlled freeze and warming was used. Cat heads or skulls were not moved in the chamber. The following three cat experiments 15-17 were performed using the freezing chamber.
{It had been made in March 1997 but had not been used in that time because of greater consumption of LN2 ( 20-30 liters of LN2 per a experiment) as compared with the old chamber (the vacuum flask had a consumption of 5-7 liters of LN2 per the like experiment). We had difficulties with LN2 in that time, though to work using the new chamber was easier than using the old one. The staple of the new chamber is a chemical glass vessel (volume 600 ml) with a copper coil for warming. I have sent you the photo where I have been photographed with the chamber.}
15. Freezing a cat head (372 g) perfused with DMF.
The cooling rates were 36.2 C/min (0 – 30), 25.2 C/min (30 – 80) for tc1 and 7.1 C/min (5 – 80) for tc2. The warming rates were 10.8 C/min (80 – 18), 7.5 C/min (18 – 0) for tc1 and 3.4 C/min (80 – 5) for tc2. The brain of the head weighed 23,0 g.
The photos 13.1 – 13.3 show the fixed brain. The upper and lower surfaces of the brain had no macrocracks and looked well (Phs. 13.1-13.2). However there were 10-12 cracks inside the brain (Ph. 13.3 mainly, in the upper part of brain; it is the upper right piece). The cracks were of smaller sizes than the cracks inside the brain of the experiment 14.
16. Freezing a glycerolized cat head (390 g). A duplication of experiment 13.
The glycerol perfusion of the cat head of this experiment was performed in norm. The cat brain weighed 18 g. The cooling rates were 1.5-1.0 C/min (0 -196) for tc1 and 1.0 C/min (5 – 196) for tc2. The warming rates were 1.5-1.0 C/min (196 – 0) for tc1 and 1.0 C/min (196 – 5) for tc2. The cat brain weighed 20.4 g.
The photos 14.1 – 14.3 demonstrate the fixed brain. The upper and lower surfaces of the brain did not contained macrocracks (Phs. 14.1-14.2). But 8-10 macrocracks were found inside the brain (Ph. 14.3). Four of them had greater sizes than the cracks inside the brain of the experiment 15 (with DMF). In comparison with the experiment 13 (with glycerol), the brain had greater numbers of cracks. But the brain was better cryopreserved as comparison with the experiment 14 (with lower glycerol concentration).
17. Freezing a cat head (485 g) perfused with DMF. A duplication of experiment 15.
The rates of freeze-warming were as the same as in the experiment 16 (about 1 C/min). The brain of the head weighed 23,0 g. The upper and lower surfaces of the brain had no macrocracks (Phs. 15.1 – 15.2). However 10-12 cracks were found inside the brain (Ph. 15.3). In the main, the cracks were met in the upper part of brain (it is the upper right piece, Ph. 15.3) as well as in the experiment 13. But some of the cracks were greater sizes than the cracks inside the brain of that experiment 13.
In general, the results of the experiments 13 (with glycerol) and 15 (with DMF) were well reproduced in the experiments 16 and 17. Thus, the joggles did not influence on degree of cracking of deeply frozen cat brains. Even the results of the experiments 13 (with the joggles) were slightly better than the results of the experiments 16 (without the joggles). Histological preparations of all the experiments 13-17 were not made because all the cat brains had the macrocracks.
In conclusion:
1. No advantage of DMF over glycerol was found in these cat experiments as well as in the sheep experiments.
2. Rubinsky’s equation needs improvement.